Surface tension diagrams
English: In physics, surface tension is an effect within the surface layer of a liquid that causes the layer to behave as an elastic sheet. It is the effect that allows insects (such as the water strider) to walk on water, and causes capillary action, for example.
- See also surface tension
-
Bouyancy
-
Scalable illustration of capillary action for large and small bore capillaries, and for positive and negative contact angles.
-
Illustration of capillary action for contact angle < 90°.
-
Illustration of concave meniscus (contact angle < 90°)
-
Illustration of convex meniscus (contact angle > 90°)
-
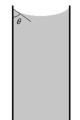
Scalable illustration of contact angle phenomenon.
-
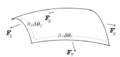
Illustration of forces on a differential patch of curved surface under tension. Used for explaining Young-Laplace equation.
-
Illustration of contact angle of droplet on surface (both contact angles less than and greater than 90° illustrated.
-
Illustration of forces at the equator of a soap bubble.
-
Diagram of mercury barometer showing negative meniscus.
-
Graph of surface tension of benzene as a function of temperature.
-
Explanatory diagram of Plateau-Rayleigh instability, which causes streams of liquid to break up into drops.
-
Illustration of forces at contact point for both contact angles less than and greater than 90°.
-
Graph of the profile of a puddle on a flat surface with contact angle of 180°.
-
Scalable diagram of droplets on a flat surface at various contact angles to illustrate the effects of surface tension and wetting.
-
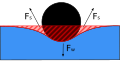
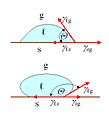
Diagram illustrating the forces acting on a needle (or other cylindrical object) floating on surface tension layer of a liquid.
-
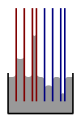
Illustration of molecular layers of surface molecules for a tiny droplet (left) and a flat surface (right).
-
Diagram of two soap bubbles sharing a common surface. Illustrates that the three surfaces must always join at 120° angle.
-
Diagram showing the force environments for molecules in a liquid, shown for both a surface molecule and for an interior molecule.